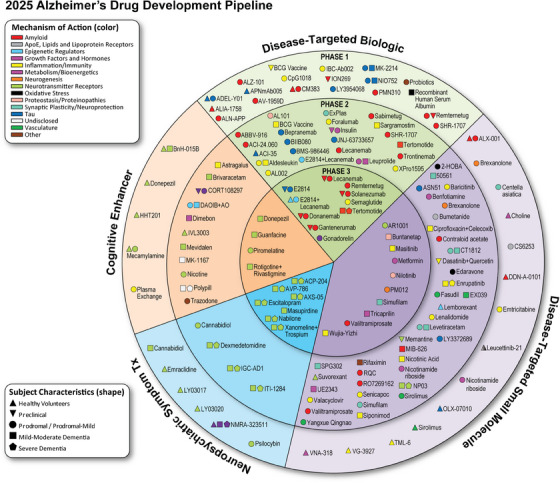
| Small Molecule Drug | Mechanism of Action | Study Phase |
| AR1001 | Nuerotransmitter Receptors | 3 |
| Buntanetap | Proteostasis | 3 |
| Mastinib | Inflammation/Immunity | 3 |
| Metformin | Metabolism/Bioenergetics | 3 |
| Niolimib | Proteostasis | 3 |
| PMO12 – herbal formula | Neurogenesis | 3 |
| Simufilam ( failed phase 3 studies) | Epigenetic regulator | 3 |
| Tricaprilin | Metabolism/Bioenergetics | 3 |
| Valiltramiprosate | prevents oligomerization of Amyloid Beta | 3 |
| Wujia Yitzhi | inflammation/Immunity | 3 |
| ACAT 1 inhibitors | 3 | |
| β‐Caryophyllene | Ameliorates STZ‐Induced Alzheimer’s Disease‐Like Conditions in Rats via Modulation of Brain‐Derived Neurotrophic Factor, Synaptic Plasticity, and Neuroinflammation – Rathod – 2025 | |
| Damona Pharmaceuticals – DPX- 101 | (PAM) that targets the α5-GABA-A receptor | 3 |
| Biological Drug | ||
| Trontinemab ( Brain Shuttled gantenerumab ) – Roche | anti amyloid + anti Transferrin Receptor FCGamma2/1 | |
| Lecanemab- | anti amyloid antibody | |
| Semaglutide | anti brain inflammation | |
| Gonadorelin | Growth factor/hormone | |
| Donanemab – | anti amyloid antibody | |
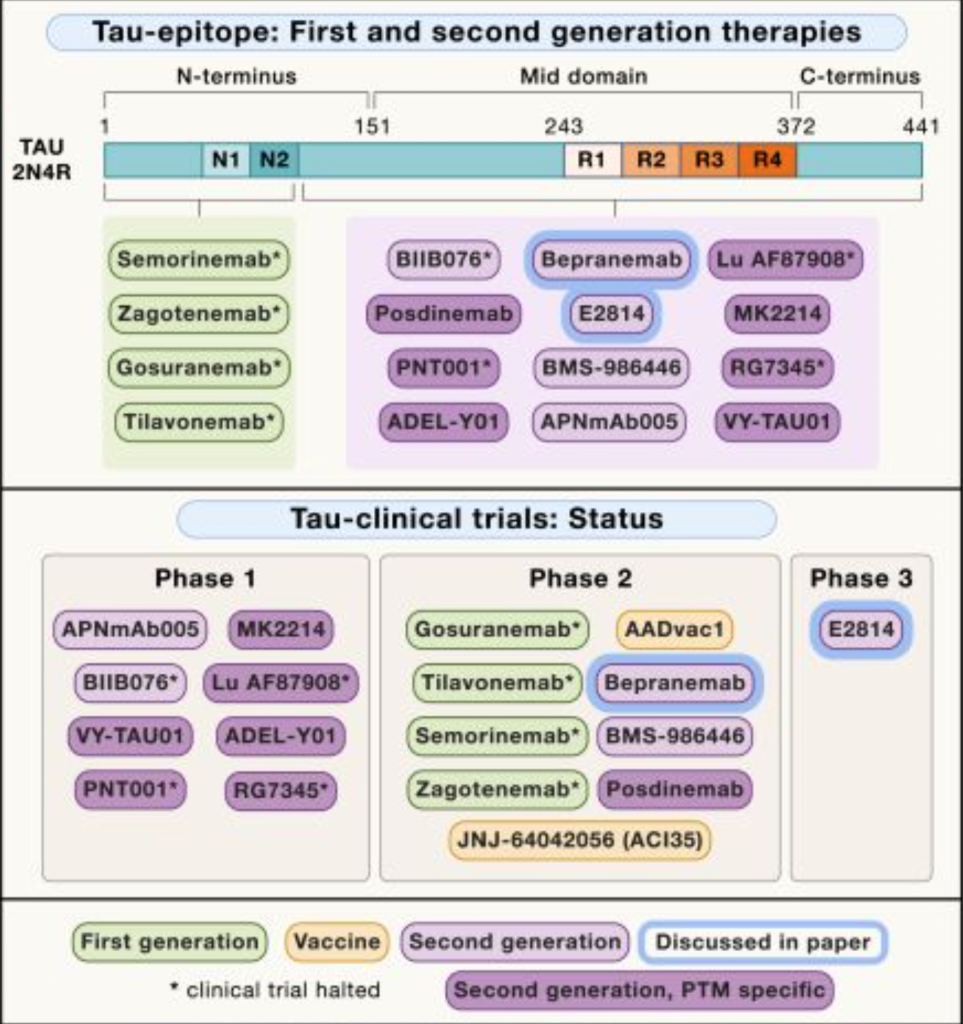
E2814 | Tau | |
Gantenerumab | anti amyloid antibody | |
| Terlomotide is a fragment derived from the human telomerase reverse transcriptase (hTERT) enzyme, specifically a 16–amino acid sequence (hTERT 611–626). | Amyloid | |
| Remternetug | anti-amyloid antibody developed by Eli Lilly | |
| Solanezumab | selectively targets and binds to soluble monomeric forms of amyloid-beta (Aβ) peptides in the brain – from Eli Lilly | |
| Posdinemab | anti -Tau antibody | |
| Bepranemab | anti- Tau antibody | |
| E2814 | anti -Tau Antibody | |
| BMS-986446 | Anti-Tau antibody | |
| APmAb005 | Anti -Tau Antibody | |
| MK2214 | Anti -Tau Antibody | |
| VY-Tau01 | Anti -Tau Antibody | |
| ADEL-Y01 | Anti -Tau Antibody | |
| anti-PDL1 antibody https://clinicaltrials.gov/study/NCT05551741 | Activation of Anti- Senescent Microglial Activity or Elimination of Senescent CD8 T cells | 1 |
| Cellular Therapy | ||
| Autologous NK cell expansion and activation by NKGen Biotech | Removal of pathogenic senescent cells | |
| Stem Cells – Various Types | Regeneration or replacement of pathogenic cells | |
| Reprogrammed T cells can eliminate senescent cells | Removal of pathogenic senescent cells | |
| CAR-T or NK Cell therapy | Removal of pathogenic senescent cells | |
| CAR-T with anti uPAR – Cold Spring Harbor/Memorial Kettering Cancer Institute | Removal of pathogenic senescent cells | |
| CAR-T with anti- KLRG1 = Anti Senescent CD8 TCell Therapy from Ortega Neuro Tx | Removal of pathogenic senescent cells | |
| Genetically engineered microglia with trans skull brain injection | Regeneration or replacement of pathogenic cells | |
| Gene Therapy – viral vector | ||
| KCTD20 gene inactivation in glutamate excitatory neurons | ||
| Intracisternal CSF injection of AAV vectors with ApoE2 christchurch and anti APOE4 miRNA | ||
| EV therapy | ||
| small Extracellular vesicles with protein exerkines, miRNA, AAV vectors, and/or circRNA | ||
| 40 Hz Ultrasound and Light | ||
| Cognito‘s Device |
Reducing pathogenic-senescent- memory CD8 T cells with T cell aPharesis or Senolytics ( drug, biologic, CAR-T, Activated NK, CAR-NK, EV, or gene therapy)
Biologics and small Drug Molecule
Each of the 11 grants is for approximately $1 million. The new Part the Cloud grant awardees are:
Translational Research Grant Program
- Glenn Larsen, Ph.D., Aquinnah Pharmaceuticals, Inc. Tauopathy Therapeutic AQV-8741: A Phase I Study.
- Terry Goldberg, Ph.D., Research Foundation for Mental Hygiene, Inc. at New York State Psychiatric Institute. Vortioxetine to improve synaptic connectivity.
- Luis Oskar Soto-Rojas, Ph.D., National Autonomous University of Mexico. Creatine-Augmented Exercise for Neuroprotection in Early AD patients.
- Timothy Siegert, Ph.D., Allyx Therapeutics, Inc. A Phase 2A Trial of ALX-001 (BMS-984923).
- Alireza Faridar, M.D., The Methodist Hospital Research Institute. IL-2 plus GLP-1 RA Combination Therapy to Target Inflammation in AD.
Gene Targeting Challenge
- Rita Balice-Gordon, Ph.D., Muna Therapeutics. Small Molecule TREM2 Agonism: Phase 1 and Observational Studies in Early AD.
Enable the Molecule Program
- Evan Lebois, Ph.D., Violet Therapeutics, Inc. Novel small molecule EPHB3 inhibitors to prevent AD synapse loss.
- Anindya Bhattacharya, Ph.D., Switch Therapeutics, Inc. Targeting MAPT knockdown by a unique siRNA platform to treat Alzheimer’s.
- Can Zhang, M.D., Ph.D., and Changning Wang, Ph.D., Massachusetts General Hospital. Discovery of novel epigenetic inhibitors for Alzheimer’s disease.
- Mark Heiman, Ph.D., and Adrian Noriega, M.D., Ph.D., Pramana Pharmaceuticals Inc. IND readiness for PRM914 a Novel Oral Gut-Brain treatment for Alzheimer’s.
- Judith Kelleher-Andersson, Ph.D., Neuronascent, Inc. Aged CCD Field Trial for Go-no-go Decision of NNI-362 Ph2 POC Trial.